Provide your details below to request scholarly review comments.
×
Verified Request System ®
Order Article Reprints
Please fill in the form below to order high-quality article reprints.
×
Scholarly Reprints Division ®
− Abstract
This article outlines the function of carbon nanotubes (CNTs) in the fabrication of biosensors for various applications. We explain why CNTs are employed more frequently in biosensor fabrications for different applications and why CNTs have significant potential in these areas of application by examining the differences between other members of the nano-carbon family. Furthermore, we investigated the role of these nano-carbons in the detection of different analytes in bio-sensing. This chapter focuses on recent breakthroughs in the construction of sensors and biosensors using carbon nanotubes (CNTs). Biosensors are employed in a wide range of applications, from prosthetic devices to checking food quality. Because of the unique physical features of CNTs nanomaterials for biosensing applications, including optical, structural, mechanical, and thermal capabilities, the scientific community has put in a lot of effort.
− Explore Digital Article Text
# I. INTRODUCTION
Biomolecular detection is vital in a variety of critical sectors, including health medications, clinical and infectious pharmaceuticals, food safety, homeland security, and pollution monitoring, as well as illness detection and identification and the recognition and viewing of novel drug molecules [1,2]. As a result, the development of dependable and affordable high-precision/selective technologies that enable direct and quick analysis of biomolecules in the detection of biomolecules can have an impact on human health, allowing for a healthier and more reliable existence [3]. The development and translation of innovative carbon nanomaterials are required for advancements in the field of biosensors [4]. The current COVID-19 epidemic has pushed the quest for novel biosensor materials [5]. The purpose of this paper is to look at biosensors that comprise carbon-based nanomaterials as well as their analytical figures of merit [6]. Biosensors have emerged as a viable choice for medical and biological investigation in applications such as environmental, medicinal, food security, and agricultural [7,8].
The utilization of carbon nanotubes (CNTs) and fullerenes in the construction of novel biosensors has recently piqued interest in carbon nanomaterials research [9]. The inclusion of sensor components permits the production of reliable, precise, and rapid biosensors due to the better capabilities of CNTs. Because of their outstanding properties in the scientific world, carbon nanotubes have spawned a plethora of studies and crucial applications. Chemical sensors, field emission materials, catalyst carriers, electronics, contemporary healthcare applications, and energy storage applications are all possible uses for CNTs [10]. CNTs have several features that make them ideal for chemical and biological sensing because of their unique structure and nanoscale size. Electrochemical sensors for food safety, optical sensors for heavy metal detection, and field-effect devices for virus detection are just a few of the uses for carbon nanotube-based sensors [11]. Large surface area to volume ratios and aspect ratios, strong electrical conductivities, greater chemical stability, and luminous characteristics are among these qualities [12]. CNTs must be purified and functionalized with bio-recognition components to make use of this nanomaterial's unique capabilities in biosensor fabrication. CNTs must be purified and functionalized with biorecognition components to make use of this nanomaterial's unique capabilities in biosensor fabrication [13]. The bio-sensing interface's design is the most difficult aspect of biosensor development. It must take into account both the functionalization and transduction processes. Food safety, health monitoring, counter-terrorism, and virus detection are all possible uses for CNT-based sensors. Even though carbon nanotubes have been reported [14,15] for three decades, few examples of CNT-based sensors have reached the market. Figure 1 shows the CNTs nanomaterials based on various types of biosensors [Ref].
The biomedical application range of CNTs is very broad [16]. There are still obstacles to the widespread use of CNT-based sensors in the real world [17], especially related to the integration of CNT sensing elements into analytical instruments and industrial manufacture. In pristine form it is like wrapped sheet of graphene and generally not solvable in most of the solvent. In some solvable solvent, it forms some bundled structure. It produces bit limitations over its usage.
CNTs' chirality can affect their electrical characteristics, and controlling this chirality can be challenging [18]. CNTs and CNT-based devices face challenges of uniformity and repeatability as a result of this. Small impurities or flaws can substantially alter the characteristics of CNTs, therefore yields and purity are also crucial, and however, these difficulties will influence SWCNTs more than MWCNTs [19]. CNTs must be synthesized with dependable control of physical and electrical characteristics in a highly repeatable way to be feasible for scale manufacturing of analytical devices [20]. Purification and subsequent functionalization of the product are required in all of the major synthesis processes.
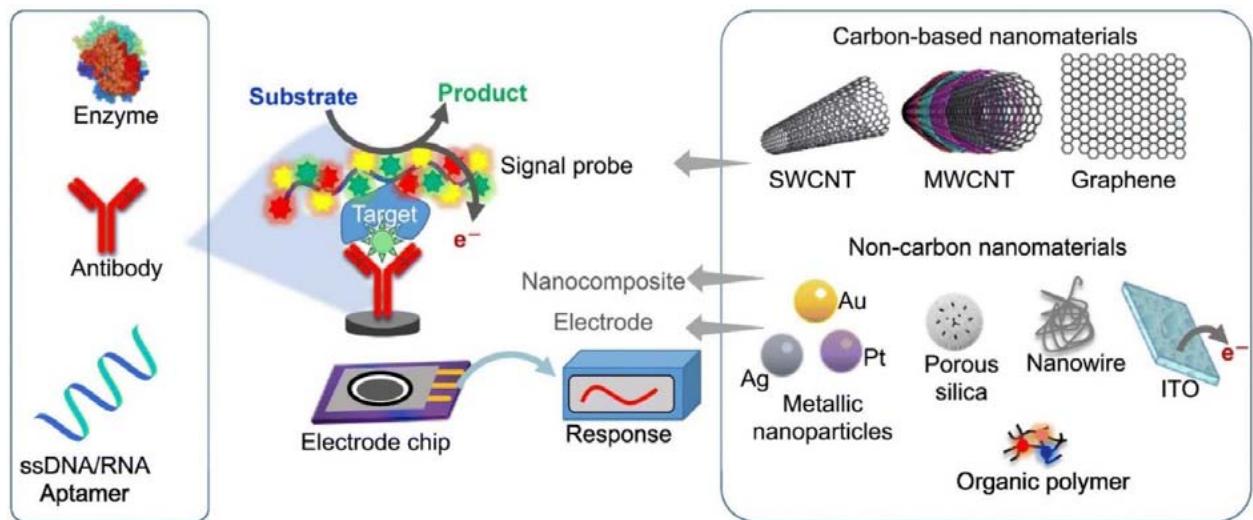
Figure 1: Schematic illustration of the CNTs nanomaterials based biosensor. This figure has been adapted/ reproduced from ref. 83 with permission from Springer, copyright 2020
Carbon nanotubes (CNTs) are hollow carbon structures having one or more walls, a nanometer-scale diameter, and a comparably longer length. They have a well-ordered arrangement of carbon atoms connected by $\mathfrak{sp}_2$ bonds, making them the stiffest and strongest fibers yet discovered [21]. Their advantage over other nanomaterials comes from a unique mix of electrical, magnetic, optical, mechanical, and chemical characteristics, all of which hold tremendous potential for a variety of applications, including biosensing [22]. Functionalized carbon nanotubes can infiltrate individual cells and cross biological barriers such as cell membranes. This property, as well as the method by which CNTs are internalized and released from cells, is of great interest for biological and, in particular, intracellular biosensing applications [23].
# II. STRUCTURE OF CARBON NANOTUBES (CNTs)
In 1991, Iijima [24] published the first report on multi-walled carbon nanotubes (MWCNTs) and their synthesis, based on findings from an arc evaporation process for the nanotubes determines the diameter of an MWCNT. CNTs are classified into two categories, SWCNTs and MWCNTs each of which is characterized by a distinct model in which Separate sheets of graphene manufacture of C60 fullerenes [ref.19]. CNTs are cylindrical in shape and have nanometer-scale diameters and micrometer-scale lengths. CNTs can be compared to a rolled-up sheet of graphene in terms of structure. Single-walled carbon nanotubes (SWCNTs) and multi-walled carbon nanotubes (MWCNTs) are the two primary forms of carbon nanotubes (CNTs). The numbers of concentric are stacked in concentric cylinders, one within the other, in the Russian Doll paradigm. In the Parchment model, a single sheet of graphene is rolled around itself in the same way as a parchment scroll or roulade cake is wound around itself as shown in figure 2.
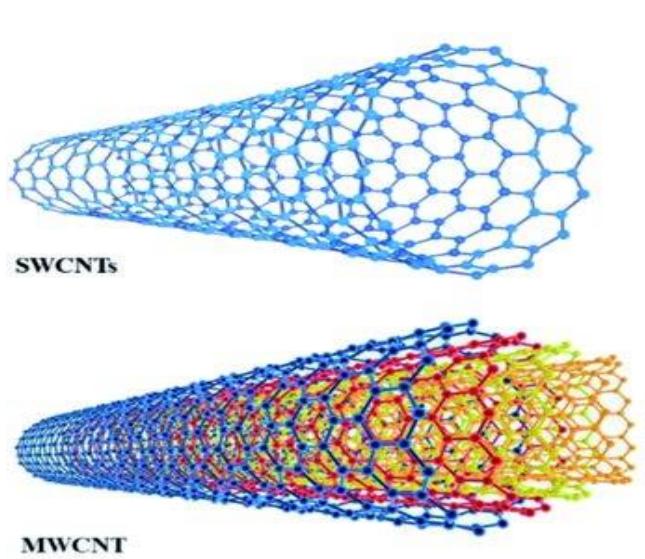
Figure 2: Nanostructures of SWCNTs and MWCNTs, fullerenes and a carbon nano-onion (CNO). Reproduced with permission from ref. 60, copyright 2008, RSC
Multiple-walled carbon nanotubes (MWCNTs) are a particular class of carbon nanotubes with shapes and characteristics comparable to SWCNTs. They are also more chemically stable, making them more resistant to the rather severe chemical procedures required for functionalization [1] diameter of less than $2\mathrm{nm}$ in most cases. SWCNTs are sometimes referred to as a single big molecule, whereas MWCNTs are a mesoscale graphite system with diameters ranging from 2 to $200\mathrm{nm}$.
The geometrical structure and C-C bonding of SWCNTs are responsible for their outstanding mechanical capabilities. One of the strongest bonds in nature is the carbon-carbon bond in graphite's base plane. The Young modulus and tensile strength of SWCNTs with tiny diameters are both high. This suggests SWCNTs are overly stiff and difficult to bend. SWCNTs are, in fact, quite adaptable. The sidewalls of these tubes are form of a hexagonal lattice of carbon atoms, comparable to the atomic planes of graphene, and one-half of a fullerene-like molecule is normally capped at both ends. SWNTs have the simplest form and may be seen as a single graphene sheet wrapped up in a numerous directions that shown in figure 3. The structure of a nanotube may be simply characterized by its chiral vector, which is determined by the chiral indices and is based on the orientation of the tube axis about the hexagonal lattice (n,m). The geometric arrangement of carbon atoms at the cylinder seams is used to classify SWNTs. While the majority of SWNTs are chiral (m ≠ n), a few have armchair $(\mathrm{m} = \mathrm{n})$ or zigzag $(\mathrm{m} = 0)$ layouts. They are flexible enough to be bent like a pipette without breaking. SWCNTs can show metallic or semiconductor behavior depending on their diameters and the chiral angle between hexagons and the tube axis, but MWCNTs can transport up to $10^{9}$ A cm $^2$ , are always metallic, and have no bandwidth.
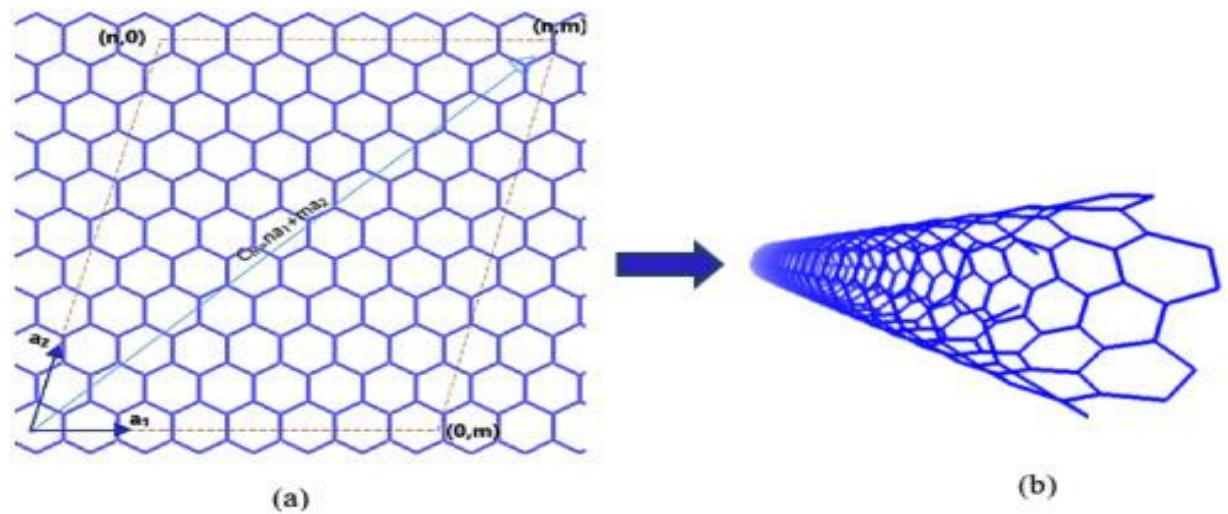
Figure 3: Rolling a graphene sheet in multiple directions to generate an SWCNT is a creative process [Ref]
CNTs are the ideal choice for enhancing bio-sensing instruments because of their exceptional properties, particularly their outstanding electrical properties such as electron mobility (100,000 cm^{2}/Vs), and field-effect mobility (79,000 cm^{2}/Vs), and electrical conductivity (104 S/cm).
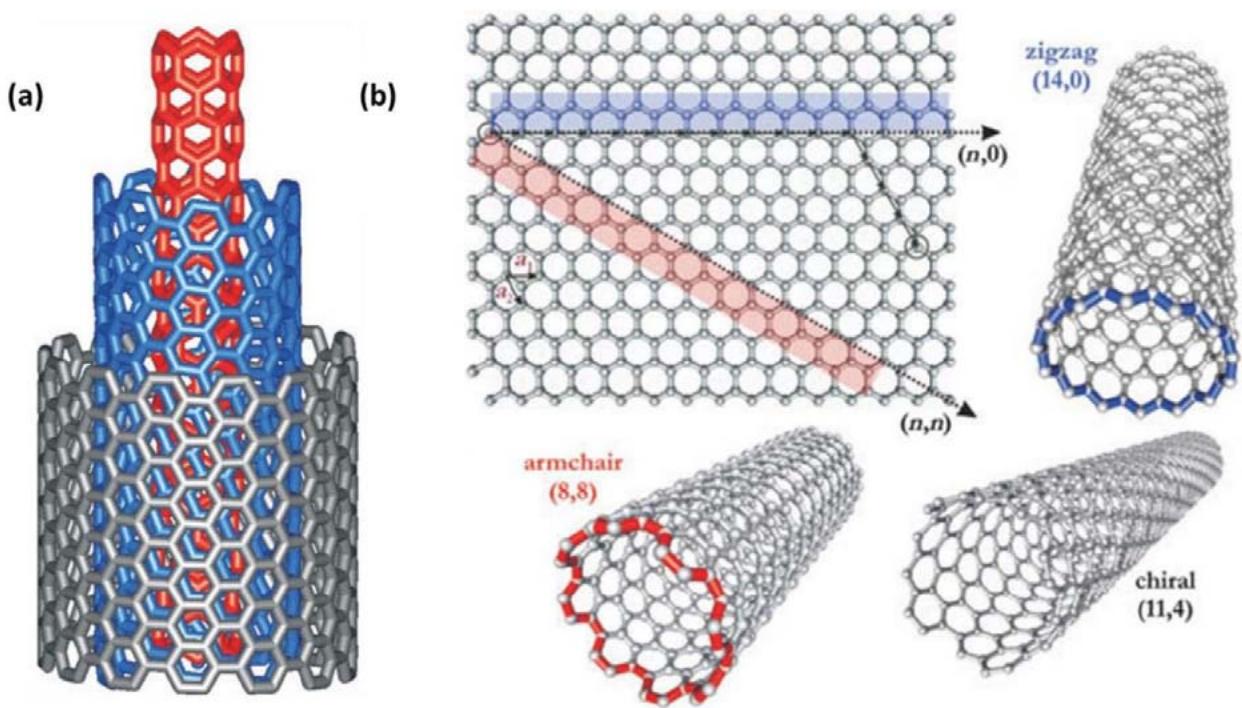
Figure 4: (a) A multi-walled carbon nanotube structure composed of three shells of varying chirality. (b) A graphene sheet is rolled up, resulting in three distinct forms of CNTs. Reproduced with permission from [25]. Copyright (2004) John Wiley and Sons
# 2.1 Functionalization of Carbon Nanotubes (CNTs)
CNTs have a number-severing qualities and benefits, however, dispersion caused by the CNTs' high surface energy acts as a road-block to future advancement [25]. Because carbon nanotubes have a high hydrophobicity, they are insoluble in water and other common solvents. As a result, CNTs must be functionalized depending on the application to increase their solubility and other functional qualities. The functionalization of carbon nanotubes has become a need for the easy production of nano-devices. Non-covalent and covalent surface modifications are two of the most popular types of functionalization. The fundamental benefit molecules stay unchanged, making subsequent modification exceedingly difficult. CNTs' solubility and compatibility can be considerably altered by direct covalent functionalization. The functionalization of carbon nanotubes has sparked a lot of attention since their carbon chemistry allows for a wide range of changes. Minimizing aggregation, enhancing their water solubility, and improving their biocompatibility have all been investigated. Because carbon nanotubes have a vast surface area, they can accommodate a large number of functional groups. Chemical, physical, and electrochemical procedures are the most prevalent methods for functionalizing carbon nanotubes. Biosensors are being developed by these functionalizing carbon nanotubes [26].
# 2.1.1 Chemical Functionalization
The chemical functionalization of CNTs is still problematic due to the difficulties in carboxyl, acyl, and hydroxyl groups are the most widely described. This procedure, which is usually reproducibly generating a well-ordered molecular architecture of the electrode surface under mild experimental conditions. A variety of techniques have been developed to change and improve the functioning of carbon nanotubes. The application of acid-based oxidization for the introduction of oxygen-containing combinations is successful in enhancing oxygen-containing functional groups on both functional groups such as carried out using nitric or nitric/sulphuric acid the ends and sidewalls of CNTs. Oxidative therapy, on the other hand, might have a wide range of results. Even though oxidative treatment increases the hydrophilic character of CNTs, controlling the degradation of the electrical, optical, and electrochemical characteristics of CNTs is typically challenging. Defect sites can be introduced during oxidative treatment, resulting in undesirable chemical and physical characteristics. The inclusion of these oxygen-containing functional groups enables the CNTs to be further modified. These can be used as a starting point for adding or converting a new functional group. Strong acid treatments utilized in chemical methods for CNT alterations were shown to be successful not only in adding oxygen-containing functional groups but also in removing metallic catalysts. However, the time required for acid oxidation of CNTs is restricted, and this is frequently deemed undesirable in industrial applications [27].
# 2.1.2 Physical Functionalization
By adding oxygen-containing groups on the end and sidewall of CNTs, several methods for functionalizing them have been investigated. Plasma treatment has been used to graft various functional groups such as carboxylic, hydroxyl, or amine groups among these methods. A variety of plasma gases, including oxygen, air, a hydrogen-nitrogen combination, or carbon dioxide, might be utilized for this purpose. Plasma techniques are an efficient means of improving the hydrophilic character of CNTs, making biosensor construction easier by allowing biorecognition components to be immobilized. In the case of an aligned configuration of CNTs, microwave plasma treatment with carbon dioxide or nitrogen/hydrogen not only allowed the CNTs to be functionalized but also prevented aggregation, preserving the electrode surface's alignment structure. When compared to the traditional oxidative treatment in a solution, the atmospheric plasma treatment was shown to be more suited for the functionalization of CNTs with an aligned configuration [28-29].
# 2.1.3 Electrochemical Functionalization
Functionalized CNTs might be functionalized using electrochemistry under mild reaction conditions, avoiding unwanted and uncontrolled reactions. Diazonium salt reactions are a novel way to functionalize the end-tip and sidewall of carbon nanotubes. For function-arising CNTs, interesting findings have been obtained with diazonium salts; nevertheless, due to the spontaneous polymerization process of radical species created by decomposing diazonium salts, getting monolayers with a well-controlled architecture remains hard. CNTs can be anchored to an electrode surface using the diazonium salt technique. It was shown that a well-organized assembly of CNTs may be achieved, resulting in a straight forward method for changing electrode surfaces using CNTs. Aryl diazonium salt derivatives have the benefit of being able to be applied to a variety of surfaces, including CNTs with metal nanoparticles [30].
# 2.2 Methods for Preparation of CNTs
The use of CNTs in biosensors is becoming increasingly common. As a result, the industry's need for CNTs has grown increasingly critical. Arc discharge, laser ablation, and chemical vapor deposition are some of the synthesis methods used.
# 2.2.1 Arc Discharge
The arc discharge is one of the most used ways of preparing carbon nanotubes, and it was initially used to generate CNTs by Iijima in 1991 [31]. The idea of this approach is that CNTs are deposited on the graphite cathode in a vacuum reactor under the action of the current. The cathode is a bulky graphite rod, whereas the anode is a fine graphite rod; the spacing between the two graphite electrodes has remained within $1\mathrm{mm}$ and the arc has remained stable. To prevent CNT oxidation, the vacuum reactor is filled with a specified amount of inert gas, such as helium or argon. CNTs are frequently synthesized under certain loading conditions, such as high current (50-120 A), high voltage (20-30 V), high temperature $(>3000^{\circ}\mathrm{C})$ and low pressure (50-700 bar). The graphite and metal catalyst of the anode graphite rod is vaporized and consumed continuously during the arc discharge process under a high-temperature plasma; CNTs are deposited on the graphite cathode following the action of metal catalysts. The synthesis of CNTs is dependent on the selection and management of process parameters such as arc voltage, current, inert gas pressure, and electrode cooling speed. The selection of metal catalysts for CNT synthesis is equally critical since it affects the preparation's efficiency. The arc discharge is one of the most well-established synthetic processes for SWCNTs, and it is also one of the most widely employed. Varied catalysts for the production of SWCNTs result in different diameter distributions, according to research [32].
# 2.2.2 Laser Ablation
Smalley and his colleagues developed laser ablation as a simple and effective new technology based on arc discharge. To begin, a piece of graphite is put in a vacuum furnace filled with an inert gas (for example, helium) and heated to a high temperature. The high temperature will be created quickly utilizing a high-energy laser beam to irradiate the metal catalyst and carbon atoms of targeted graphite, prompting them to evaporate, and then the carrier gas will transport these carbon molecules and catalyst particles to the high-temperature zone. To make CNTs, the carbon atom clusters are finally placed on the collector. The most notable benefit of laser ablation is the excellent purity and low defect rate of the CNTs generated. Furthermore, the yield of CNTs might be as high as $70\%$ to $90\%$ . However, because this technique of growing CNTs has a high initial investment cost and significant energy consumption, it is challenging to use in commercial manufacturing [17,33].
# 2.2.3 Chemical Vapor Deposition (CVD)
CVD is a thermal dehydrogenation reaction that requires a transition metal catalyst, and it is one of the most extensively used and well-explored methods for the synthesis of CNTs. First, the carbon source gas and carrier gas enter the reaction chamber at a temperature of 600–1000 °C, where the gas is decomposed to form carbon atoms on a coated catalyst substrate at a high temperature, and eventually, CNTs are produced. Co, Fe, Ni, Cu, Cr, Mo, and their alloys are often used as transition metal catalysts. Carbon gases such as methane, ethanol, ethylene, acetylene, and benzene are commonly used as carbon sources. This may aid in lowering the temperature necessary for gaseous hydrocarbon breakdown into carbon and hydrogen. Carbon supply, gas flux, carrier gas flux, growth temperature, and catalyst concentration are the most important CVD process factors. CVD is split into the matrix technique and floating catalyst method based on the loading and dispersion methods of the catalyst. CVD has the benefit that CNTs can grow directly on the substrate, and the equipment is simple and inexpensive. It is worth mentioning that this technology may be used for mass manufacturing indefinitely [34].
# 2.3 Different CNT-based Biosensor's Working Principles
Clark's 1962 description of the enzyme electrode inspired the notion of a CNT-based biosensor; CNT-based biosensors are made up of two parts: a biologically sensitive element and a transducer. The biological sensitive element is a CNT functionalized with biomolecules or receptors, such as proteins, oligo- or polynucleotides, microbes, or even complete biological tissues. For testing and detection, the transducer converts analyte concentrations to other observable physical signals such as currents, absorbance, mass, or acoustic variables. CNT-based biosensors will be divided into two groups depending on the interactions between analytes and biologically sensitive materials, which will be explored in the following sections: chemical and physical. The CNTs-field transistors, which have better characteristics, will be emphasized after this section [13, 35].
# 2.4 Biosensors based on Carbon Nanotubes
The first biosensor was produced in 1906, but Clark et al. invented the real biosensor in 1956 and became the father of biosensors. He then created an amperometric enzyme electrode for glucose sensing in 1962. Based on the interactions between the analytes and the biological sensitive materials, CNT-based biosensors were divided into two categories (chemical and physical).
# 2.4.1 Electrochemical CNT Biosensors
Various signal amplification methodologies using CNTs have been explored to develop ultrahigh sensitivity biosensors. Because of the reactive assemblies on the outer surface of CNTs, they have fascinating electrochemical characteristics. CNTs have been hailed as a promising material for electrochemical biosensors because of their ability to improve electron transfer. Electrochemical biosensors function on the premise of converting/transferring a biological event into an electrochemical signal using a two or three-electrode electrochemical cell (reference, working, and counter electrode). High sensitivity, cheap production cost, rapid response, ease of operation, and prospective mobility distinguish CNT-based electrochemical biosensors as outstanding materials in biomedical applications [36].
# 2.4.2 Electrochemical Enzyme Sensors
Since the creation of the first enzyme-based biosensor for the detection of blood glucose, electrochemical biosensor technology, and research have evolved tremendously. Enzyme-based electrochemical biosensors are a subclass of chemical sensors that combine enzyme specificity and selectivity with electrochemical sensing's high sensitivity. The common enzymes used in biosensor development of oxidoreductases, catalyze oxidation-reduction events involving the production of electro-active substances. One of the most difficult aspects of designing enzyme-based electrochemical biosensors has been achieving effective electrical communication between immobilized enzymes and the electrode surface. The benefit of this generation is that the concentration of diffusive electroactive species and the measured current are directly proportional. Biosensors that do not require the use of a mediator are referred to as mediator's biosensors. Electrochemical biosensors based on enzymes are a type of chemical sensor that combines the specificity and selectivity of enzymes with the high sensitivity of electrochemical sensors. Several studies using enzymes such as glucose oxidase horseradish peroxidase, lactate oxide, and malate dehydrogenase have been published. Oxidoreductase-based electrochemical biosensors including oxidoreductases are enzymes that catalyze oxidation/reduction reactions by making electron transfer from reductants (electron donors) to oxidants easier (electron acceptors). Electrons are eventually transferred to convert ADP to ATP in most oxidation processes. Non-protein chemical substances or metallic ions that are essential for enzyme action are known as cofactors. Coenzymes are complex organic cofactors. Several coenzymes have been found, and vitamins are the most common cofactors. $\mathrm{X}^{-} + \mathrm{Y} \rightarrow \mathrm{X} + \mathrm{Y}^{-}$ . Where, $\mathrm{X}$ is the reductant and $\mathrm{Y}$ is the oxidant, is the typical reaction. They work with inorganic and organic cofactors to act on diverse functional groups like --CHO, -CHOH, CH2-NH2, -CH=NH, or -CH2-CH2-. Electron transfer reactions between biomolecules catalyzed by oxidoreductase (especially NAD(P)H) play critical roles in the metabolism and catalysis conversion of various materials in plants and liver tissues, including adenosine diphosphate (ADP) to ATP cycles, ethanol to acetaldehyde, glucose to gluconolactone, pyruvate to lactate, and so on. Furthermore, NAD(P)H's reducing capacity is necessary for additional metabolic pathways that are catalyzed by various enzyme and coenzyme cofactors [37].
# 2.4.3 Electrochemical Immunosensors
Immunosensors are condensed analytical biosensor devices based on ligand affinity, in which immunochemical reaction interaction and molecular recognition between antigen and antibodies are connected to a transducer for selective detection of various proteins. Immunosensors are devices that make use of antibody-antigen binding interactions. A sensor surface can be immobilized with either the antibody or the matching antigen, and the binding of the corresponding member of the pair, producing a stable complex, can be detected using several methods. By combining SWCNT forests with enzymes and horseradish peroxidase, the electrochemical immunosensor displayed extremely low and enhanced cancer-related detection of interleukin-6, a multifunctional cytokine. Because of the very particular nature of their bonding with their respective antigens, antibodies are commonly utilized in biosensing applications, and there are several instances of CNT-immunosensors [38].
Furthermore, MWCNTs-based impedimetric immunosensors adorned with gold nanoparticles have been effectively used to detect cancer cells in serum, particularly in breast cancer patients. The combination of highly conductive ionic liquid and MWCNTs, as well as gold nanoparticles, improved the sensitivity of the proposed immunosensor while also improving biocompatibility and amplification power [39].
# 2.4.4 Electrochemical DNA Sensors
Because of its powerful and very specific hybridization, DNA is an appealing bio-recognition component for sensors. Medicine and healthcare, food safety, and counterterrorism are just a few of the applications that DNA sensors might be used for. Covalent binding of the nucleic acids' amine terminal to CNT-functionalized electrodes is a typical way of immobilizing nucleic acids onto CNT-functionalized electrodes.
The sensing element of a DNA biosensor is a nucleic acid, such as single-stranded DNA (ssDNA) or double-stranded DNA (dsDNA). It has been discovered that ssDNAs adsorb strongly on CNTs, but duplex DNAs are unable to attach to CNTs in a stable manner. This one-of-a-kind characteristic has been used to meticulously build CNT-DNA biocomplexes for use in biosensing technologies for a variety of molecular targets. The SWCNT serves as a transducer, converting and amplifying DNA hybridization on Au into an electrical signal that can be measured immediately. This newly created sensing platform has proved to have considerably simpler chemistry and setup than most currently available optical and other electrochemical DNA biosensors. Furthermore, a simple and ultra-sensitive DNA biosensor based on MWCNT signal amplification and fluorescence polarization (FP) detection for monitoring the activity and inhibition of DNA methyltransferase (MTase) in a homogenous solution has been successfully created. DsDNA and restriction endonuclease was used to make the DNA probe. Restriction endonuclease without DNA MTase cleaves the FAM-labeled DNA probe [40].
# 2.4.5 Non-Biomolecule-based Electrochemical Sensors
Sensors based on molecularly imprinted polymers have gained popularity in recent years (MIPs). MIPs are antibodies that have been synthesized. Simply said, polymers are made with the target molecule attached to the polymer structure either covalently or noncovalently. While the biomolecule-based sensors discussed in the preceding sections are likely the most popular electrochemical CNT-based sensors, there are numerous more that do not use biomolecules as recognition components. CNTs are of particular importance for MIP applications because they may increase the porosity and mechanical characteristics of polymer composites while also imparting or enhancing electrical conductivity. CNTs have also been widely employed to functionalize electrode surfaces to increase the sensitivity of direct electrochemical detection for a range of analytes in combination with other nanomaterials. Noble metal nanoparticles, such as gold and silver, have been utilized to functionalize electrodes for the detection of analytes such as glucose, urea, bisphenol A, and volatile organic compounds (VoCs) alongside CNTs [41].
# 2.4.6 Optical Biosensors
The CNTs have shown great optical behavior and the feature of showing different kinds of luminescent behavior makes them an ideal optical probe for biosensing. They have shown photoluminescence, fluorescence, electroluminescence, cathodoluminescence, Raman scattering etc. The CNTs can play very important role in the fabrication of variety of optical biosensors. Specifically, the single walled carbon nanotubes [SWCNTs] are very promising one for the purpose. CNTs' luminous features may be used for biosensing applications using a range of biomolecules. Optical biosensors work on the premise of sensing photons (absorbance, reflectance, or fluorescence emissions) rather than electrons to detect and analyze interactions between target biomolecules and analytes. Optical transduction methods are extensively utilized in biosensors because they have excellent sensitivity, stability, and multiplex detection capabilities. CNTs are commonly used in fluorescence sensors and biosensors because their fluorescent characteristics are strongly reliant on their physical structure. CNTs can also be used to quench fluorescent light. Fluorophores can be used to mark bio-recognition elements like oligonucleotides. The oligonucleotides are then non-covalently linked to CNTs, quenching the fluorophores. Surface plasmon resonance (SPR) is a very sensitive optical method that is frequently used in chemical and biological detection. It uses optical stimulation to induce the creation of surface plasmon waves at the interface between metal surfaces, such as gold, and a dielectric. The fact that SWCNTs emit light between 700 and $1400\mathrm{nm}$ qualifies them as biocompatible fluorescence probes. SWCNTs have a wavelength range that permits rays to penetrate deeply through layers of skin, allowing in vivo imaging of biological tissues and organs with a dosage limit that is nearly 15 times lower. SWCNTs have also been used as NIR fluorescent markers for precise cell probing and imaging. The fluorescent characteristics of carbon nanotubes have also been used to detect COVID-19. A simple and quick fluorescent sensor for the SARS-CoV-2 spike protein was demonstrated by non-covalently binding the protein ACE2, which has a high binding affinity for the SARS-CoV-2 spike protein [42].
# 2.4.7 CNTs as a Field-Effect Transistor Biosensors
Field-effect transistors (FETs) are three-channel devices with source (S), drain (D), and gate (G) electrodes that are often employed to amplify weak signals. The voltage generated in the device is controlled by the gate electrode. Because silicon-based transistors have hit their scaling limit, CNT-based transistors are currently seen as one of the most promising possibilities to replace existing silicon FETs. With an increase in current density, the device transistor footprint encompassing all components (S, D, and G) can be lowered to less than $40\mathrm{nm}$ during the next decade. As we all know, the existence of high hysteresis in CNTs degrades their performance as a transistor, which is a key downside as an energy-efficient material. To lower conductivity, the first biosensor-based SWNTs were bound with the enzyme GOx. However, when dissolved in glucose, these semiconducting SWNTs serve as reversible $\mathsf{pH}$ sensors, implying that they may be used as an enzymatic biosensor. The FET biosensors based on N-acetyl-D-glucosamine (GlcNAc)-fSWCNTs controlled secretion (or exocytosis) of PC12 cells in real-time detection. Because CNTs have a high conductance, these networking molecules can make them more sensitive to the electrochemical concentration at the surface [43-45].
# III. FUTURE PROSPECTIVE
The physical and electrical features of CNTs have been highlighted in the preceding sections as being excellent for the construction of biosensors. To improve or exploit these properties, CNTs may be mixed with a variety of different nanomaterials to make composite materials. Food safety, health monitoring, counterterrorism, and virus detection are all possible uses for CNT-based sensors. Even though carbon nanotubes have been around for three decades, few CNT-based sensors have made it to market. There are still obstacles to the widespread use of CNT-based sensors in the real world, especially related to the integration of CNT sensing elements into analytical instruments and industrial manufacture. As the world's health systems respond to the effect of the coronavirus pandemic in the early 2020s, research interest in quick viral diagnostic techniques, as well as areas like point-of-care patient monitoring, will likely increase. CNTs have attracted a lot of attention in biomedical engineering during the last decade because of their diverse surface characteristics, size, and form. CNTs are becoming a promising material due to their inorganic semiconducting capabilities as well as their organic-stacking features. As a result, it could successfully interact with biomolecules while also responding to the light. CNT-based nanomaterials might be utilized to produce biomedical applications in the future by combining these features into a single entity. CNT-based analytical devices will undoubtedly play a prominent part in many of the approaches to these difficulties because of their potential for building biosensors with high sensitivity and quick reaction times.
# IV. CONCLUSION
CNTs' exceptional electrochemical characteristics have cleared the way for their usage as platforms for the development of a wide range of electrochemical biosensors with better analytical performance. In vivo detection with reduced cytotoxicity, high sensitivity, and long-term stability is possible using CNT-based bio-sensing for reliable point-of-care diagnostics under physiological settings. Furthermore, these applications are not limited to the medical area, but also have a wide range of uses in the food, water purification, and agricultural industries, among others. As a result, it's safe to state that CNTs' genuine bio-sensing applications are still a ways off. Although CNT-based biosensors appear promising, there are still several practical problems in terms of implementation. Biosensor manufacture, for example, often requires a precise size and helicity, but controlling the size of CNTs during production is difficult. It's also difficult to mass-produce CNTs in a cost-effective and high-purity manner, which is why the present market price of CNTs is far too exorbitant for any practical commercial application.
Generating HTML Viewer...
− Conflict of Interest
The authors declare no conflict of interest.
− Ethical Approval
Not applicable
− Data Availability
The datasets used in this study are openly available at [repository link] and the source code is available on GitHub at [GitHub link].