Provide your details below to request scholarly review comments.
×
Verified Request System ®
Order Article Reprints
Please fill in the form below to order high-quality article reprints.
×
Scholarly Reprints Division ®
− Abstract
The present study was conducted to assess the effect of temperature, pH, and salt on bacteriocin activity produced from different strains of Lactic Acid Bacteria. On the basis of cultural, biochemical, physiological and morphological identification the Lactobacillus species were identified. Four species of Lactobacillus were isolated from homemade cheese, raw milk, dosa paste, sauce, and curd. Lactobacillus species isolated from the sampled food source include; Lactobacillus brevis, Lactobacillus plantarum , Lactobacillus fermentum, and Lactobacillus casei. The bacteriocins activity towards different levels of temperature, pH and salt was recoreded.The results showed that the highest bacteriocin activity was observed at 60°C for 180 min and the lowest bacterial activity was observed at 121°C for 20 min. L.plantarum(P2) resported the maximum bacteriocin activity.The highest bacteriocins activity was reported at pH-6. Across different bacterial strains tested for pH stability, L. plantarum(P2) reported maximum bacteriocins activity. It was observed that bacteriocins produced from L. brevis reported highest salt tolerance. Across different bacterial strains tested for salt tolerence, L. brevis reported maximum bacteriocins activity. The bacteriocin produced from LAB in the current experiment show resistance to pH, heat, and different organic solvents, thus this bacteriocin opened the prospect for its utilization in preservation of food products.
− Explore Digital Article Text
# I. INTRODUCTION
Lactic Acid Bacteria (LAB) have been intensively used in food preservation and research across the globe has reported that LAB stimulates the nutrient level of different fermented foods and its derived products (Sameh, et.al., 2016; Raman, et.al., 2022). LAB improves the flavor, taste, texture, and shelf-life of different products (Korcari, et.al., 2021). The LAB creates lactic acid, which makes the environment more acidic and decreases the number of pathogenic microorganisms. LAB has the ability to create organic acids, hydrogen peroxide, diacetyl, and bacteriocins which are some of the antibacterial substances. These substances are known to lessen food deterioration and the growth or proliferation of harmful bacteria. As a result, the food industry has begun to pay more attention to the utilisation of these naturally occurring chemicals as food bio-preservative agents, which currently offers a viable alternative to chemical food preservation, particularly for ready-to-use products. (Suskovic et.al., 2010, Lappa, et.al., 2022). Bacteriocins could also be used to prepare products that aren't being sufficiently thermally sterilized at the time of their production, as there is a risk of product becoming contaminated with pathogenic microorganisms like *Listeria monocytogenes*, which has been linked to numerous outbreaks around the world (O'Sullivan et.al., 2002).
Bacteriocins are ribosomally-synthesized peptides or proteins with antimicrobial activity, produced by different groups of bacteria. Many Lactobacillus sp. produce bacteriocins with rather broad spectra of inhibition. Several LAB bacteriocins offer potential applications in food preservation, and the use of bacteriocins in the food industry can help to reduce the addition of chemical preservatives as well as the heat intensity treatments, resulting in foods that are better maintained naturally and have stronger nutritive and organoleptic qualities. This can be an alternative to satisfy the increasing consumer demands for safe, fresh-tasting, ready-to-eat, minimally-processed foods and also to develop "novel" food products (e.g. less acidic, or with a lower salt content) (Alkena et.al., 2016). The inhibitory spectrum of some bacteriocins also includes food spoilage and/or food-borne pathogenic microorganisms. In the past years, a lot of works have been aimed to the detection, purification and characterization of bacteriocins, as well as to their use in food preservation strategies. However, it's essential to report the tolerance of bacteriocins towards different physical and chemical factors of environment. Keeping in view the role of bacteriocins, the present study was conducted to assess the effect of temperature, pH, and salt on bacteriocin activity produced from different strains of Lactic Acid Bacteria.
# II. MATERIAL AND METHODOLOGY
# 2.1. Isolation and Identification of LAB
Homemade cheese, raw milk, dosa paste, sauce, curd was serially diluted and spread on MRS agar medium. Plates were incubated anaerobically for 24-48 hrs at $30^{\circ}$. When required for the isolation of specific bacteria and incubated aerobically and anaerobically at various temperatures. A total of 13 isolates of bacterial species, namely Aeromonas, Bacillus, Clostridium, Escherichia, Klebsiella, Listeria, Salmonella, Streptococcus, Staphylococcus, Enterobacter, Enterococcus, and Lactobacillus were isolated from the sampled dairy products. On the basis of cultural, biochemical, physiological and morphological identification the Lactobacillus species were identified. Four species of Lactobacillus species were identified; they include; L. plantarum (P2), L. brevis, L. plantarum (P1), L. casei, and L. fermentum. The morphological characteristics of the bacterial strain obtained from different food sources is given in Table-1.
Table-1: Morphological characteristics of the bacterial strain obtained from different food sources.
<table><tr><td>Strain code</td><td>Cell's form</td><td>Type</td><td>Colour</td><td>Motility test</td><td>Gram staining</td></tr><tr><td>L. brevis</td><td>Rod shaped</td><td>Bacilli</td><td>Yellow</td><td>Non-motile</td><td>Gram positive</td></tr><tr><td>L. plantarum</td><td>Slender rods</td><td>Coccobacilli</td><td>Yellow</td><td>Non-Motile</td><td>Gram positive</td></tr><tr><td>L. fermentum</td><td>Rod Shaped</td><td>Cocci</td><td>Creamy White</td><td>Non-Motile</td><td>Gram positive</td></tr><tr><td>L. casei</td><td>Rod-shaped</td><td>Smooth</td><td>Opaque with pigment</td><td>Non-Motile</td><td>Gram positive</td></tr></table>
# 2.2. Production of Bacteriocin
The bacteriocin production from the isolated strains of LAB species (L. plantarum (P2), L. brevis, L. plantarum (P1), L. casei, and L. fermentum) was grown in MRS broth (Hi Media Lab, Pvt Ltd. India) with $1\%$ inoculum and it was maintained at optimum culture conditions for 48 hours. The cells were removed from the growth medium after incubation by centrifugation at $15000\mathrm{rpm}$ for 15 minutes at $4^{\circ}\mathrm{C}$. The obtained cell-free supernatant was adjusted to $\mathrm{pH}6.5$ using $1\mathrm{mol/L NaOH}$ and it was used as crude bacteriocin.
# 2.3. Purification of Bacteriocin
Two methods were used for this purpose; Ammonium salt precipitation and ion-exchange chromatography (Yang et al., 2018).
# 2.3.1. Ammonium Salt Precipitation
Ammonium salt precipitation, various concentrations of Ammonium Sulphate (10, 20, 30, 40, 50 and $60\%$) was added to $10\mathrm{ml}$ of crude bacteriocin in different test tubes, precipitated for $24\mathrm{h}$ at $4^{\circ}\mathrm{C}$. Then the obtained mixture of bacteriocin was centrifuged at $10,000~\mathrm{rpm}$ for $10\mathrm{min}$ and then the obtained precipitate was further resuspended in $25\mathrm{ml}$ of $0.05\mathrm{M}$ of Potassium Phosphate buffer (\mathrm{pH} 7) (Yang et al., 1992). Further dialysis was followed in dialysis bags against 2 litres of the same buffer.
# 2.3.2. Ion-Exchange Chromatography
The dialysate was used for purification by cation exchange column (DEAE cellulose column) and elution was performed by using a linear gradient from citrate phosphate buffer ranging from pH 2.6 to 7.0 (Macher et al., 1980). The bacteriocin titer was assessed (Todorov et al., 2004) of eluted samples.
# 2.4. Effect of Temperature, pH, and Salt on Bacteriocin Activity
In order to assess the impact of temperature, the purified bacteriocin activity was tested by incubating it at different temperatures between $60^{\circ}\mathrm{C}$ to $121^{\circ}\mathrm{C}$ and the residual activity was tested after 20 minutes, 40 minutes, 120 minutes, and 150 minutes. To examine the effect of $\mathsf{pH}$, the purified bacteriocin activity was tested by incubating it at different $\mathsf{pH}$ between 2 to 12 with sterile 1 mol/L NaOH or 1 mol/L HCl. Further the effect of salt on the purified bacteriocin activity was observed by taking $400\,\mu L$ of the purified bacteriocin, and it was incubated at $37^{\circ}\mathrm{C}$ with NaCl concentrations ranging from 10 to $40\%$. (Merck, Germany). Well-diffusion assay was used to measure the activity.
# III. RESULTS AND DISCUSSION
Four species of Lactobacillus were isolated from homemade cheese, raw milk, dosa paste, sauce, and curd. Lactobacillus species isolated from the sampled food source include; Lactobacillus brevis, Lactobacillus plantarum, Lactobacillus fermentum, and Lactobacillus casei. Table-1 and Table-2 shows the morphological, physiological and biochemical attributes of L. brevis, L. plantarum, L. fermentum, and L. casei isolated from cheese, raw milk, dosa paste, sauce, and curd. Lactobacillus brevis is a facultative anaerobic, hetero-fermentative species that is commonly found in fermented foods, such as beer, wine, and vegetables. It is a Gram-positive, non-spore-forming, rod-shaped bacterium that can occur singly, in pairs, or in short chains. The cells typically measure $0.7 - 1.0 \mu \mathrm{m}$ in width and $2.0 - 4.0 \mu \mathrm{m}$ in length, with rounded ends. Lactobacillus plantarum is a Gram-positive, non-motile, non-spore-forming, micro-aerophilic, and mesophilic bacterium. The cells are straight rods with rounded ends, measuring $0.9 - 1.2 \mu \mathrm{m}$ in width and $3.0 - 8.0 \mu \mathrm{m}$ in length, and can occur singly, in pairs, or in short chains. Some strains of L. plantarum can possess true catalase and manganese-containing pseudo-catalase activities, as well as nitrate- and hematin-dependent nitrite reductases. Lactobacillus fermentum is a Gram-positive, non-spore-forming, rod-shaped bacterium that is commonly found in fermented foods and the human gastrointestinal tract. The cells are typically $0.5 - 0.8 \mu \mathrm{m}$ wide and $2.0 - 4.0 \mu \mathrm{m}$ long, with rounded ends, and can occur singly, in pairs, or in short chains. Lactobacillus casei is a Gram-positive, non-spore-forming, rod-shaped bacterium that is widely used in the production of fermented dairy products and as a probiotic. The cells are typically $0.5 - 0.8 \mu \mathrm{m}$ wide and $1.0 - 10.0 \mu \mathrm{m}$ long, with rounded ends, and can occur singly, in pairs, or in short chains.
Table-2: Physiological and Biochemical characteristics of Isolated Lactobacilli.
<table><tr><td>Characteristics</td><td>Raw milk</td><td>Curd</td><td>Cheeses</td><td>Sauce</td><td>Dosa Batter</td></tr><tr><td>Species</td><td>L. plantarum P1</td><td>L. plantarum P2</td><td>L. casei</td><td>L. brevis</td><td>L. fermentum</td></tr><tr><td>Morphology</td><td>Cream colony Gram +ve Rod</td><td>Cream Colony Gram +ve Rod</td><td>Cream Colony Gram +ve Rod</td><td>White colony Gram +ve Rod</td><td>White colony Gram +ve Rod</td></tr><tr><td>15 °C only</td><td>+</td><td>+</td><td>+</td><td>+</td><td>-</td></tr><tr><td>45 °C only</td><td>-</td><td>-</td><td>-</td><td>-</td><td>-</td></tr><tr><td>15 and 45 °C only</td><td>+</td><td>+</td><td>+</td><td>+</td><td>+</td></tr><tr><td>Acid and gas from glucose</td><td>-</td><td>-</td><td>-</td><td>+</td><td>+</td></tr><tr><td>NH3 from Arginine</td><td>-</td><td>-</td><td>-</td><td>+</td><td>+</td></tr><tr><td>Arabinose</td><td>-</td><td>-</td><td>+</td><td>+</td><td>+</td></tr><tr><td>Cellobiose</td><td>+</td><td>+</td><td>-</td><td>-</td><td>+</td></tr><tr><td>Mannitol</td><td>+</td><td>+</td><td>-</td><td>-</td><td>+</td></tr><tr><td>Mannose</td><td>+</td><td>+</td><td>+</td><td>-</td><td>+</td></tr><tr><td>Melibiose</td><td>+</td><td>+</td><td>-</td><td>-</td><td>+</td></tr><tr><td>Raffinose</td><td>+</td><td>+</td><td>+</td><td>-</td><td>+</td></tr><tr><td>Ribose</td><td>+</td><td>+</td><td>+</td><td>+</td><td>+</td></tr><tr><td>Salicin</td><td>+</td><td>+</td><td>+</td><td>-</td><td>+</td></tr><tr><td>Lactose</td><td>-</td><td>-</td><td>-</td><td>+</td><td>+</td></tr><tr><td>Rhamnose</td><td>-</td><td>-</td><td>-</td><td>-</td><td>-</td></tr><tr><td>Sorbitol</td><td>-</td><td>-</td><td>-</td><td>-</td><td>+</td></tr><tr><td>Xylose</td><td>-</td><td>-</td><td>-</td><td>+</td><td>+</td></tr><tr><td>Trehalose</td><td>-</td><td>-</td><td>-</td><td>-</td><td>+</td></tr></table>
Due to the fact that all bacteriocin was created during the pre- and early exponential growth phases and reached a maximum level at late stationary phase, the test isolates synthesis of bacteriocin demonstrated secondary metabolic kinetics. According to certain accounts, bacteriocins are created at all times during the experimental growth phase rather than just in the late logarithmic or early stationary phases. The process of producing bacteriocin was optimised by taking into account many parameters, including carbon and nitrogen sources, pH, temperature, and salt content. The optimum parameter was then determined using an arbitrary unit. The addition of glucose (2.0%) increased the amount of bacteriocin that could be produced, while the addition of other carbon sources had no effect or had a negative impact on production. For the nitrogen source, the medium's maximum production was achieved by mixing Tryptone, yeast extract, and meat extract together. While extremely alkaline and acidic pH did not enable the formation of bacteriocin, maximum activity in composition medium was attained at initial pH ranging from 6-8, and the optimal temperature was 30°C. The bacteriocin was created at its highest level under the ideal conditions, and once it had been purified, it could be employed right away as a bio-preservative. The manufacturing of bacteriocins can be optimised to produce them more cheaply, which could eliminate the need to add chemical preservatives altogether or at least limit their use.
Effect of Temperature, PH, and Salt on Bacteriocin activity Produced from Different Strains of Lactic Acid Bacteria (LAB)
Table-3: Effect of Temperature on the activity (AU/μl) of bacteriocin produced by the isolated strains of LAB species.
<table><tr><td rowspan="2">Temperature</td><td colspan="2">L. plantarum(P1)</td><td colspan="2">L. plantarum(P2)</td><td colspan="2">L. casei</td><td colspan="2">L. brevis</td><td colspan="2">L. fermentum</td></tr><tr><td>ZI (mm)</td><td>AU/μl</td><td>ZI (mm)</td><td>AU/μl</td><td>ZI (mm)</td><td>AU/μl</td><td>ZI (mm)</td><td>AU/μl</td><td>ZI (mm)</td><td>AU/μl</td></tr><tr><td>60°C for 150 min</td><td>11.0</td><td>110</td><td>15.0</td><td>150</td><td>11.0</td><td>110</td><td>13.0</td><td>130</td><td>10</td><td>100</td></tr><tr><td>80°C for 120 min</td><td>9.0</td><td>90</td><td>13.0</td><td>130</td><td>10.0</td><td>100</td><td>8.0</td><td>80</td><td>7.0</td><td>70</td></tr><tr><td>100°C for 40 min</td><td>8.0</td><td>80</td><td>12.0</td><td>120</td><td>5.0</td><td>50</td><td>7.0</td><td>70</td><td>0</td><td>0</td></tr><tr><td>121°C for 20 min</td><td>8.0</td><td>80</td><td>12.0</td><td>120</td><td>0</td><td>0</td><td>0</td><td>0</td><td>0</td><td>0</td></tr></table>
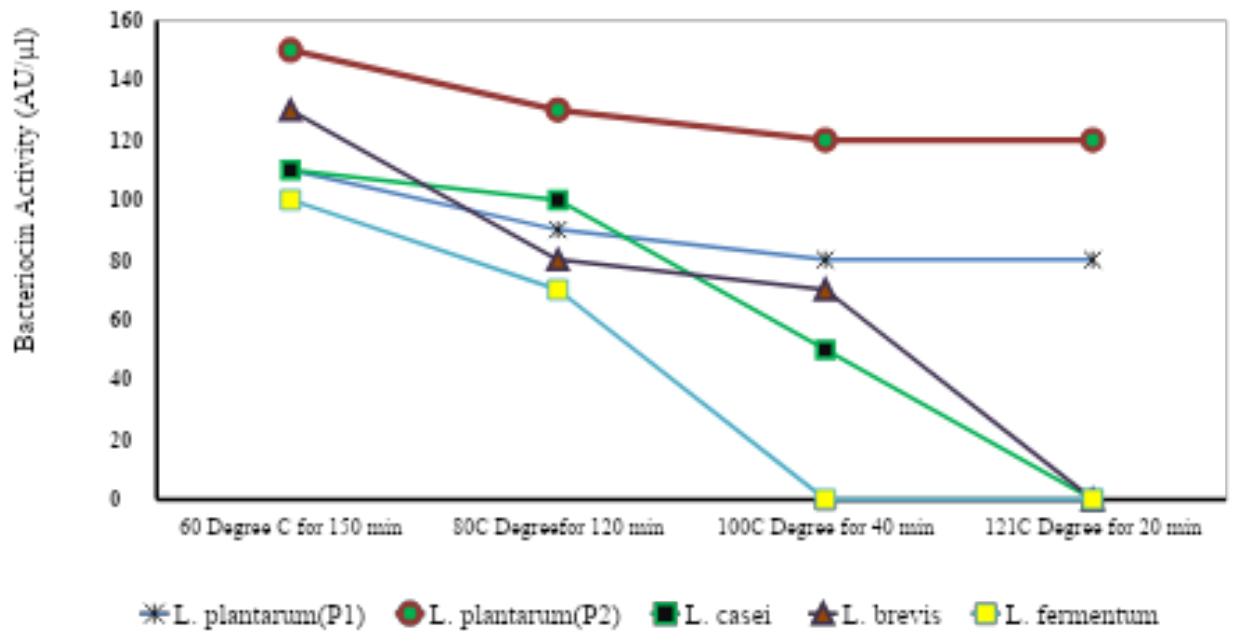
Fig-1: Thermal stability of Bacteriocin after exposure at different temperature treatment
The optimum condition for maximum bacteriocin activity was demonstrated by culturing the bacteria among various thermal ranges. Purified bacteriocin was exposed to various heat treatments: $60^{\circ}\mathrm{C}$ for 180 min, $80^{\circ}\mathrm{C}$ for 120 min, $100^{\circ}\mathrm{C}$ for 40 min and $121^{\circ}\mathrm{C}$ for 20 min (Table-3, Fig-1). The results showed that the highest bacteriocin activity was observed at $60^{\circ}\mathrm{C}$ for 180 min and the lowest bacteriocin activity was observed at $121^{\circ}\mathrm{C}$ for 20 min. Across different bacterial strains L. plantarum(P2) recorded the maximum bacteriocin activity followed by L. brevis, L. plantarum(P1), L. casei, and L. fermentum. The results report that with the increase in the temperature there was a decrease in the activity $(\mathrm{AU} / \mu \mathrm{l})$ of bacteriocins. The L. plantarum(P2) showed the highest thermal stability that the other bacteriocins in terms of the temperature to which they were exposed.
Effect of Temperature, PH, and Salt on Bacteriocin activity Produced from Different Strains of Lactic Acid Bacteria (LAB)
Table 4: Effect of pH on the activity (AU/μl) of bacteriocin produced by the isolated strains of LAB species.
<table><tr><td rowspan="2">pH</td><td colspan="2">L. plantarum(P1)</td><td colspan="2">L. plantarum(P2)</td><td colspan="2">L. casei</td><td colspan="2">L. brevis</td><td colspan="2">L. fermentum</td></tr><tr><td>ZI (mm)</td><td>AU/μl</td><td>ZI (mm)</td><td>AU/μl</td><td>ZI (mm)</td><td>AU/μl</td><td>ZI (mm)</td><td>AU/μl</td><td>ZI (mm)</td><td>AU/μl</td></tr><tr><td>2</td><td>10</td><td>100</td><td>15</td><td>150</td><td>11</td><td>110</td><td>13</td><td>130</td><td>09</td><td>90</td></tr><tr><td>4</td><td>14</td><td>140</td><td>17</td><td>170</td><td>12</td><td>120</td><td>16</td><td>160</td><td>10</td><td>100</td></tr><tr><td>6</td><td>15</td><td>150</td><td>19</td><td>190</td><td>13</td><td>130</td><td>18</td><td>180</td><td>12</td><td>120</td></tr><tr><td>8</td><td>11</td><td>110</td><td>18</td><td>180</td><td>09</td><td>90</td><td>16</td><td>160</td><td>07</td><td>70</td></tr><tr><td>10</td><td>05</td><td>50</td><td>18</td><td>180</td><td>05</td><td>50</td><td>15</td><td>150</td><td>00</td><td>00</td></tr><tr><td>12</td><td>00</td><td>00</td><td>17</td><td>170</td><td>00</td><td>00</td><td>15</td><td>150</td><td>00</td><td>00</td></tr></table>
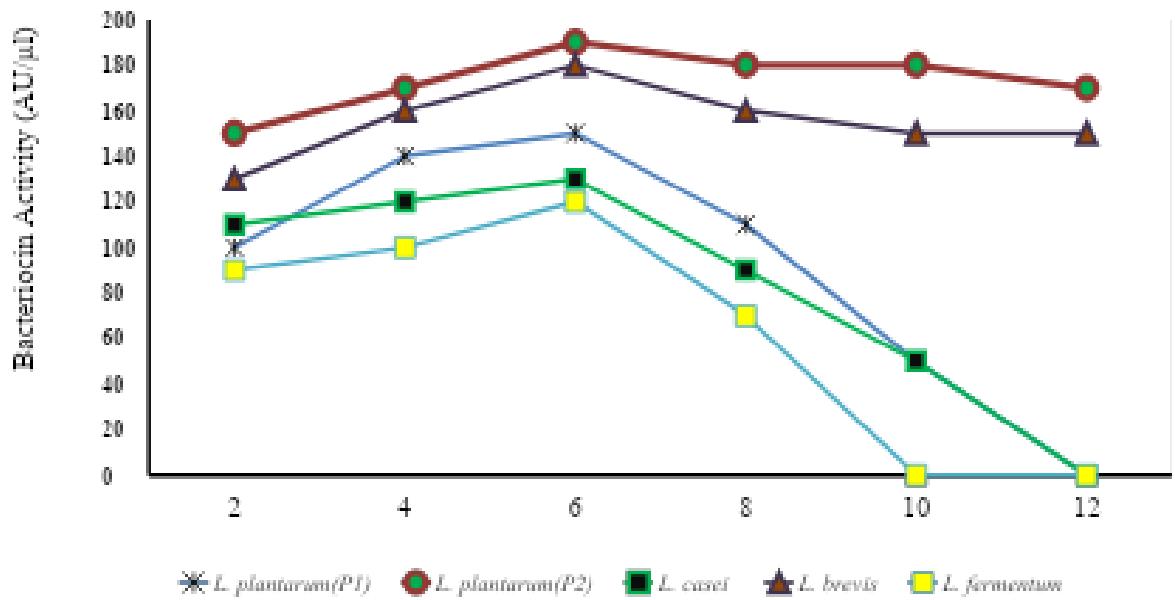
Fig-2: pH stability of Bacteriocins produced from different LAB species after exposure at different pH
The influence of pH on the activity of bacteriocins. Stability of pH of bacteriocins was observed at pH range of 2.0 to 12.0. It was observed that the bacteriocins activity report maximum stability between pH 4-6. The highest bacteriocins activity was reported at pH-6 (Table-4). Across different bacterial strains tested for pH stability, L. plantarum(P2) reported maximum bacteriocins activity followed by L. brevis, L. plantarum(P1), L. casei, and L. fermentum (Table-5, Fig-2). L. plantarum (P2) showed was reported to be the highest pH stable of bacteriocins after exposure at different pH. The overall results report that the pH affects the bacteriocins activity significantly.
Table-5: Effect of Salt on the activity (AU/μl) of bacteriocin produced by the isolated strains of LAB species.
<table><tr><td rowspan="2">Salt Conc.</td><td colspan="2">L. plantarum(P1)</td><td colspan="2">L. plantarum(P2)</td><td colspan="2">L. casei</td><td colspan="2">L. brevis</td><td colspan="2">L. fermentum</td></tr><tr><td>ZI (mm)</td><td>AU/μl</td><td>ZI (mm)</td><td>AU/μl</td><td>ZI (mm)</td><td>AU/μl</td><td>ZI (mm)</td><td>AU/μl</td><td>ZI (mm)</td><td>AU/μl</td></tr><tr><td>10%</td><td>12.0</td><td>120</td><td>19</td><td>190</td><td>11.0</td><td>110</td><td>20.0</td><td>200</td><td>11.0</td><td>110</td></tr><tr><td>20%</td><td>10.0</td><td>100</td><td>18</td><td>180</td><td>11.0</td><td>110</td><td>19.0</td><td>190</td><td>9.0</td><td>90</td></tr><tr><td>30%</td><td>8.0</td><td>80</td><td>16</td><td>160</td><td>7.0</td><td>70</td><td>18.0</td><td>180</td><td>8.0</td><td>80</td></tr><tr><td>40%</td><td>0.0</td><td>00</td><td>16</td><td>160</td><td>5.0</td><td>50</td><td>18.0</td><td>180</td><td>6.0</td><td>60</td></tr></table>
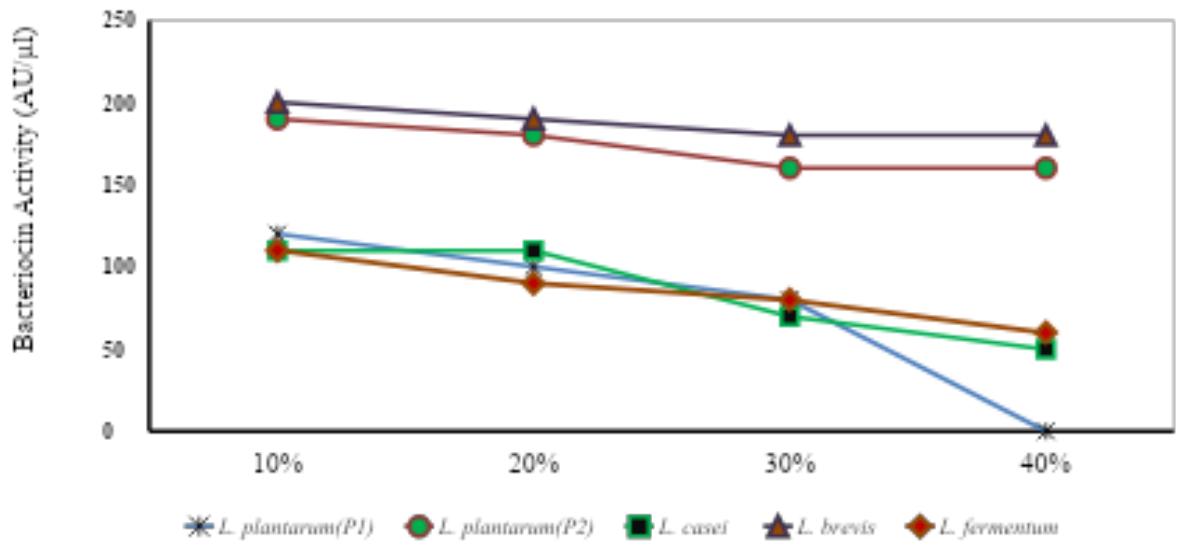
Fig. 3: Salt tolerance of Bacteriocins produced from different LAB species.
$400\mu \mathrm{l}$ of the purified bacteriocin were incubated at $37^{\circ}\mathrm{C}$ with NaCl concentrations ranging from 10 to $40\%$ . (Merck, Germany). Well diffusion assay was used to measure the activity. It was observed that bacteriocins produced from L. brevis reported highest salt tolerance. The tolerance by bacteriocin activity was recorded lowest by L. casei. Across different bacterial strains tested for salt tolerance, L. brevis reported maximum bacteriocins activity followed by L. plantarum(P2), L. plantarum(P1), L. fermentum, and L. casei (Table-5, Fig-3). The overall results show that the bacteriocin activity was highest at $20\%$ salt concentration and lowest at $40\%$ salt concentration.
# IV. DISCUSSION
The present study examines the effect of temperature, pH and salt on the bacteriocin activity which were produced from Lactobacillus species isolated from Raw milk, Dosa, Curd, Sauce and Cheese. The Lactobacillus species were identified on the basis of morphological, biochemical and physiochemical attributes. Out of these five Lactobacillus species, two were identified as $L.$ plantarum and coded as P1 and P2, respectively, due to their different potential in bacteriocin activity, and the other three were identified as $L.$ brevis, $L.$ casei, and $L.$ fermentum. The bacteriocins produced by the Lactobacillus species in this investigation were subject to different levels of temperature, pH and salt to determine the stability of the bacteriocins for their future applications.
The Lactobacillus bacteriocin isolated from different samples raw milk, dosa paste, curd, sauce and cheese have thermal stability at $60^{\circ}\mathrm{C}$ for 150 min. The results showed that the highest bacteriocin activity was observed at $60^{\circ}\mathrm{C}$ for 150 min and the lowest bacterial activity was observed at $121^{\circ}\mathrm{C}$ for 20 min. This is significant if the bacteriocin is to be utilized as a food preservative because many food preparation processes require a heating stage. The mechanism of heat-stability of LAB bacteriocins has been reported earlier for Plantaricin S (Jimenez-Diaz et al., 1990), Plantaricin A (Daeschel et al., 1985), Plantaricin 149 (Kato et al., 1994), Plantaricin SA6 (Rekhif et al., 1995), Plantaricin 423 (Van-Reenen, 1998), Plantaricin C19 (Audisio, 1999), Pentocin TV35b (Okkers et al., 1999), L. brevis oG1 (Ogunbanwo et al., 2003), and lactocin RN78 (Mojgani and Amirinia, 2007). Because we noted heat stability of L. plantarum P2 bacteriocin, the conclusions of the present study are consistent with the findings of the papers cited above. This bacteriocin belongs to the category of bacteriocins with low molecular weight that are heat stable and retain their activity after heating at $121^{\circ}\mathrm{C}$ for 60 min. The bacteriocin is superior in processed foods when high heat is used because of this property. The thermal stability of other bacteriocins was not good. Additionally, Andersson (1986) noted a decrease in activity following a 15-minute heat treatment at $121^{\circ}\mathrm{C}$.
The test isolates regarding bacteriocin's activity was also pH-dependent. The bacteriocin produced by L. brevis, L. plantarum(P1), L. plantarum(P2), L. casei, and L. fermentum showed pH stability at acidic pH (2-6). However only L. brevis and L. plantarum(P2) retained its activity at alkaline pH (8-12). L. fermentum have completely lost their activity from pH 10 and L. brevis lost its complete activity at pH 12 respectively. The overall bacteriocin activity was reported highest between pH 4-6. This was also demonstrated by Reddy et al. (1984) and Abdel-Bar et al. (1987) in their studies of two bacteriocins, bulgarican and lactobulgarian, isolated from L. bulgaricus, which had the highest activity and stability at pH 2.2 and 4.0, respectively, against a variety of pathogenic and spoilage bacteria.
In particular, pH 6 was found to have increased antibacterial activity in the bacteriocin generated by $L.$ brevis, L. plantarum(P1), L. plantarum(P2), L. casei, and L. fermentum. This might be because bacteriocins' increased net charge at low pH makes it easier for the molecules to pass through the cell wall. At lower pH levels, bacteriocin solubility may also rise, aiding the diffusion of bacteriocin molecules. This concurred with earlier studies that demonstrated that the presence of NaCl enhanced the antimicrobial action of bacteriocins like nisin, leucocin F10, enterocin AS-48, and others. A modest quantity of NaCl also inhibited nisin action (Bouttefroy et al., 2000). Additionally, sodium chloride reduced the antilisterial efficacy of the antibiotics acidocin CH5 (at 1-2%; Chumchalova et al., 1998), lactocin 705, leucocins 4010 (at 2.5% NaCl; Hornbaek et al., 2004), pediocin, (at 6.5% NaCl; Jydegaard et al., 2000), curvacin (Vaerluyten). The ionic interactions between bacteriocin molecules and charged groups involved in bacteriocin binding to target cells may be interfered with by sodium chloride, which has a protective effect (Bhunia et al., 1988.) In addition, sodium chloride may cause bacteriocin structural alterations or modifications to the target cell's envelope (Lee et al., 1993) or changes in the cell envelope of the target organisms (Jydegaard et al., 2000).
The bacteriocin produced from LAB in the current experiment show resistance to pH, heat, and different organic solvents, thus this bacteriocin opened the prospect for its utilization in preservation of food products. It can also confirm that these LAB strains can be used as starting cultures for microorganisms and as bio-preservatives in a variety of fermented foods. The most typical bacteria for food fermentation and preservation are LAB. Their significance is primarily related to their safe metabolic activity as they consume accessible sugar while growing in food to produce organic acids and other metabolites (Gomashe et al. 2014).
# V. CONCLUSION
Lactic Acid Bacteria (LAB) can produce a range of antimicrobial compounds, such as organic acids, hydrogen peroxide, and bacteriocins, which can effectively inhibit the growth of various microorganisms. Four species of Lactobacillus were isolated from homemade cheese, raw milk, dosa paste, sauce, and curd. Lactobacillus species isolated from the sampled food source include; Lactobacillus brevis, Lactobacillus plantarum, Lactobacillus fermentum, and Lactobacillus casei. The bacteriocins activity towards different levels of temperature, pH and salt was recorded. The characterization of Bacteriocins from test isolates were studied in order to take advantage of their potential and make them viable candidates for deployment as a safe and effective biological preservative in the future. Across different LAB species the bacteriocins produced from L. plantarum report distinctive and comprehensive heat stability, pH instability, and salt tolerance, can be exploited as a biopreservative to increase the food items' hygienic and safety (especially processed foods). The use of Lactobacillus species as natural biopreservatives in food products is a promising approach to enhance food safety and extend shelf-life, thereby reducing the reliance on synthetic preservation methods.
# Conflict of Interest
The authors have no conflict of interest regarding the publication of this manuscript
# Plagiarism and AI Content
The author declares that Plagiarism of the manuscript is below $10\%$ similarity level, and $0\%$ AI content detected.
Generating HTML Viewer...
− Conflict of Interest
The authors declare no conflict of interest.
− Ethical Approval
Not applicable
− Data Availability
The datasets used in this study are openly available at [repository link] and the source code is available on GitHub at [GitHub link].