Provide your details below to request scholarly review comments.
×
Verified Request System ®
Order Article Reprints
Please fill in the form below to order high-quality article reprints.
×
Scholarly Reprints Division ®
− Abstract
Polyphenols, known for their strong antioxidant properties, combat oxidative stress, enhance cell viability, and protect against free radical damage. These bioactive compounds hold immense potential for functional food and cosmetic innovations, supporting overall health and promoting skin resilience, offering exciting opportunities in nutraceutical and cosmetic product development. This study evaluates the bioactive properties and potential applications of Collas-Co™, a high-polyphenol ingredient with promising antioxidant activity. The total phenolic content (TPC) of Collas-Co™ was determined to be 58.05 ± 0.33 mg GAE/g. Its antioxidant capacity, assessed via ORAC assay, revealed a remarkably high scavenging activity of 15,221 ± 409 µmol TE/100g. Furthermore, the DPPH assay demonstrated superior radical scavenging activity, with Collas-Co™ achieving 85.67% inhibition at 2.5 mg/mL, outperforming gallic acid by approximately 15%. Cytotoxicity studies using primary human dermal fibroblasts indicated that Collas-Co™ exhibited dose-dependent effects. At lower concentrations (0.39 mg/mL), it enhanced cell viability to 112.94%, suggesting potential protective or stimulatory properties. Moderate concentrations (0.78–6.25 mg/mL) showed tolerability, while higher concentrations (>12.50 mg/mL) resulted in cytotoxicity, with an IC50 of 50.50 ± 2.07 mg/mL. These findings highlight Collas-Co™ as a potent antioxidant with moderate cytotoxicity, suitable for functional foods and cosmetic applications.
− Explore Digital Article Text
# I. INTRODUCTION
The increasing global prevalence of oxidative stress-related conditions, such as cardiovascular diseases, neurodegenerative disorders, diabetes, and certain cancers, highlights the critical need for effective antioxidant strategies. Oxidative stress results from an imbalance between reactive oxygen species (ROS) production and the body's ability to neutralize these harmful molecules, leading to cellular damage, inflammation, and aging [1]. Polyphenols, a class of naturally occurring bioactive compounds, have garnered significant attention due to their potent antioxidant properties. These compounds act as free radical scavengers, protect cellular structures, and inhibit oxidative damage, making them valuable in addressing chronic diseases and promoting overall health [2-3].
Collas-Co™, a novel polyphenol-rich ingredient, has demonstrated exceptional potential for health applications due to its high total polyphenol content (TPC). Polyphenols are not only indicators of antioxidant potential but are also associated with cellular protective mechanisms that support skin health, delay aging, and mitigate oxidative damage. While TPC provides preliminary insights into bioactivity, comprehensive evaluations of antioxidant capacity and cellular safety are crucial to validate its efficacy and safety for human applications. Techniques such as the Oxygen Radical Absorbance Capacity (ORAC) and DPPH radical scavenging assays are widely regarded as robust methods to assess the radical-scavenging ability of antioxidants [4-5].
Furthermore, cytotoxicity studies are essential to determine the safety of polyphenol-rich ingredients, particularly when intended for nutraceuticals, functional foods, or cosmetics. Fibroblasts, key cellular components of the skin, play a crucial role in maintaining extracellular matrix integrity and repairing oxidative damage. Investigating the impact of Collas-Co™ on fibroblast viability provides valuable insights into its cytotoxicity profile, cellular safety, and potential for regenerative applications. This is particularly important as cellular health is directly linked to skin vitality and anti-aging benefits [6].
This study focuses on evaluating the antioxidant activity and cellular safety of Collas-Co™, with a specific emphasis on its polyphenol-rich composition and radical-scavenging efficacy. Additionally, a comparative analysis with gallic acid, a well-known antioxidant, highlights Collas-Co™'s relative effectiveness. The findings aim to establish Collas-Co™ as a safe, multifunctional ingredient suitable for addressing oxidative stress, promoting cellular health, and supporting its application in functional food, nutraceutical, and cosmetic industries.
# II. MATERIALS AND METHODS
# 2.1 Materials
Collas-Co™ was procured from Beyond Laboratory (Thailand) Co., Ltd., Bangkok, Thailand, while all chemicals used in the analysis were of analytical reagent (AR) grade.
# 2.2 Total Polyphenol Content Analysis
The total polyphenol content (TPC) was modified according to Singleton [7]. A 1000 ppm gallic acid solution was prepared by dissolving $0.25\mathrm{g}$ of gallic acid (Sigma-Aldrich, Seelze, Germany) in $1.5\mathrm{mL}$ of ethanol, followed by dilution to a final volume of $250\mathrm{mL}$ with deionized water. From this stock solution, standard solutions were prepared at concentrations of 10, 25, 50, 100, 200, and $250\mathrm{ppm}$. Separately, $25\mathrm{g}$ of sodium carbonate (Sigma-Aldrich, Seelze, Germany) was dissolved in $100\mathrm{mL}$ of warmed deionized water, stirred until fully dissolved, filtered, and cooled to room temperature. Additional sodium carbonate was added incrementally to encourage crystallization; if crystallization did not occur, the solution was stored in a refrigerator for future use. For the Collas-Co™ solution, $1.00\mathrm{g}$ of sample was dissolved in $100~\mathrm{mL}$ of boiling deionized water with stirring for approximately 10 minutes. The solution was filtered, and the residue was re-extracted with an additional $100~\mathrm{mL}$ of boiling deionized water. The filtrates from both extractions were combined and diluted to a final volume of $250~\mathrm{mL}$ in a volumetric flask.
For the assay, $0.5\mathrm{mL}$ of each gallic acid standard, sample solution, and a deionized water blank were transferred to separate tubes. To each tube, 4.3 mL of deionized water, 0.2 mL of Folin-Ciocalteu's phenol reagent, and 0.5 mL of saturated sodium carbonate were added sequentially. The mixture was vortexed, followed by the addition of another 4.3 mL of deionized water, and vortexed again. The tubes were incubated in the dark at room temperature for 45 minutes, and the absorbance was measured at 765 nm to determine phenolic content. TPC was calculated using the equation of the standard curve: $\mathrm{y} = 0.0048\mathrm{x} + 0.0111$ with a coefficient of determination $(\mathrm{R}^2 = 0.9986)$. Results were expressed as mg GAE/g (milligrams of gallic acid equivalents per gram).
# 2.3 Antioxidant Activities Analysis
# 2.3.1 Oxygen Radical Absorbance Capacity (ORAC)
The ORAC value was measured following the procedure outlined by Kuti [8], with slight modifications. The reaction mixture consisted of $1.7\mathrm{mL}$ of $75~\mathrm{mM}$ phosphate buffer $(\mathrm{pH}7.0)$, $100~\mu \mathrm{L}$ of R-phycoerythrin (R-PE, $3.4\mathrm{mg/L})$, $100~\mu \mathrm{L}$ of $320~\mathrm{mM}$ $2,2^{\prime}$-azobis(2-amidinopropane) dihydrochloride (AAPH), and $100~\mu \mathrm{L}$ of the extract. Phosphate buffer served as the blank, and Trolox was used as the standard. The total volume of the mixture was $2\mathrm{mL}$, placed in a $10~\mathrm{mm}$ fluorometer cuvette, and preincubated at $37^{\circ}\mathrm{C}$ for 15 minutes. The reaction was initiated by adding AAPH, and fluorescence was recorded every 5 minutes at $570~\mathrm{nm}$ (emission) and $540~\mathrm{nm}$ (excitation) using a fluorometer (Sequoia-Turner model 450, USA), until fluorescence decreased to less than $5\%$ of the initial value. Each sample was tested in triplicate.
The ORAC values, expressed as Trolox equivalents (TE) per 100 gram, were calculated based on the area under the fluorescence decay curve. ORAC was determined using the standard curve equation $\mathrm{C} = \mathrm{K} \times \mathrm{A}$, where $\mathrm{K} = 203.65 \mu/\mathrm{mL}$, $\mathrm{A} =$ absorbance, and the coefficient of determination was $\mathrm{R}^2 = 0.9993$.
# 2.3.2 DPPH Radical Scavenging Activity
DPPH radical scavenging assay was conducted following the procedure outlined by Sadeer & Siddeega [9-10]. The DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging assay was conducted to assess the antioxidant activity of Collas-Co™ and the standard gallic acid. A 0.1 mM DPPH solution was prepared in methanol, while test samples (Collas-Co™) were diluted to final concentrations of 1.0, 0.5, 0.25, 0.125, 0.063, 0.031, 0.016, and 0.008 mg/mL using 0.5 M acetic acid. Similarly, gallic acid was prepared at concentrations ranging from 10 to 0.078 mg/mL in methanol. In a 96-well plate, 100 μL of DPPH solution was added to each well, followed by 100 μL of the sample or standard solution. Methanol and DPPH alone served as control (A0) and blank (A2), respectively. The plate was incubated at room temperature for 20 minutes in the dark to allow the reaction between the antioxidants and DPPH radicals. Absorbance was measured at 517 nm using a microplate reader. The percentage of DPPH radical inhibition was calculated using the formula: Inhibition (%) = [1 - (A_1 - A_2) / A_0] × 100
where
$$
\begin{array}{l} \mathrm {A} _ {\mathrm {o}} = \text {absorbance of control (without sample)} \\ \mathrm {A} _ {1} = \text {absorbance of test (Sample + DPPH)} \\ \mathrm {A} _ {2} = \text {absorbance} \\ \end{array}
$$
The $\mathrm{IC}_{50}$ value, indicating the concentration required to inhibit $50\%$ of DPPH radicals, was determined from the dose-response curve plotting inhibition percentage against concentration. This approach effectively quantified the antioxidant potential of the samples.
# 2.4 Cytotoxicity Test On Primary Human Dermal Fibroblast By WST-1 Assay
# 2.4.1 Cell Cultivation
Human dermal fibroblast cells (HDFn) were cultured in Dulbecco's Modified Eagle Medium (DMEM) supplemented with $10\%$ Fetal Bovine Serum (FBS) and $1\%$ Antibiotic/Antimycotic solution. Cells were seeded at an initial density of $2 \times 10^{5}$ cells per well in a 96-well culture plate and incubated at $37^{\circ} \mathrm{C}$ in a $\mathrm{CO}_{2}$ incubator with $5\% \mathrm{CO}_{2}$ for 24 hours. After incubation, cell growth and morphology were observed under an inverted microscope to ensure optimal conditions for subsequent testing or assays.
# 2.4.2 Cytotoxicity Test
Cytotoxicity test assay was conducted following the procedure outlined by Kamiloglu and Yin [11-12]. Sample solutions were incubated with cells in a 96-well culture plate, with the test sample Collas-Co™ applied at various concentrations. The negative control group consisted of cell culture media (DMEM), while the positive control group used $0.10\%$ sodium dodecyl sulfate (SDS). Cells were incubated at $37^{\circ}\mathrm{C}$ with $5\% \mathrm{CO}_{2}$ in a cell culture incubator for 24 hours.
After incubation, the cells were washed with phosphate-buffered saline (PBS), and $10~\mu \mathrm{L}$ of WST-1 solution was added to $100~\mu \mathrm{L}$ of DMEM in each well. The plates were re-incubated under the same conditions for an additional 30 minutes, after which absorbance was measured at a wavelength of $450~\mathrm{nm}$. The resulting color intensity correlated with the number of viable cells.
The absorbance values (OD) were used to calculate the percentage of cell viability using the equation:
Cell viability $(\%) =$ (Absorbance (OD) of cells in the test group x 100)/Absorbance (OD) of cells in the negative control group)
This method provided a quantitative assessment of cell viability under different treatment conditions.
# III. RESULT AND DISCUSSION
# 3.1 Total Polyphenol Content
The total phenolic content (TPC) of Collas-Co™ is summarized in Table 1. The TPC of Collas-Co™ was determined to be $58.05 \pm 0.33$ mg GAE/g. This value indicates that Collas-Co™ contains significantly higher TPC compared to medium and dark roasted coffee from Colombia, which contain $24.28\mathrm{mg}$ GAE/g and $21.41\mathrm{mg}$ GAE/g, respectively, as well as strawberry, which contains $22.37\mathrm{mg}$ GAE/g [13-14].
Although the relatively high TPC of Collas-Co™ suggests the presence of active compounds, this measure alone provides only preliminary insights into its biochemical potential. Phenolic content is a useful indicator of bioactive components; however, further investigation into its antioxidant activity is crucial to assess the functional benefits of Collas-Co™. Antioxidant activity, such as that measured through ORAC and DPPH provides a more comprehensive understanding of the compound's potential to neutralize oxidative stress and its applicability in various health-promoting contexts.
Table 1: Total polyphenol content and ORAC scavenging activity of Collas-Co™
<table><tr><td>Sample</td><td>Total Polyphenol Content (mg GAE/g)</td><td>ORAC Scavenging Activity (μmol TE/100g)</td></tr><tr><td>Collas-Co™</td><td>58.05 ± 0.33</td><td>15221 ± 409</td></tr></table>
* GAE: Gallic acid equivalent, TE: Trolox equivalent
# 3.2 Antioxidant Activities
# 3.2.1 Oxygen Radical Absorbance Capacity (ORAC)
The Oxygen Radical Absorbance Capacity (ORAC) assay is a widely used method to evaluate the antioxidant capacity of foods and other substances. This assay measures the scavenging activity of antioxidants against free radicals, particularly peroxyl radicals, by assessing the inhibition of oxidation of a fluorescent probe such as fluorescein. The ORAC method provides insight into the radical scavenging potential of compounds, using parameters such as the area under the fluorescein decay curve (AUC) and lag time to quantify antioxidant capacity effectively [15]. Furthermore, the proper selection of calibrator compounds is critical to ensure unbiased estimation of antioxidant activity, as highlighted by Carvalho [15].
For Collas-Co™, the ORAC scavenging activity was determined to be $15,221 \pm 409 \mu \mathrm{mol} \, \mathrm{TE} / 100 \, \mathrm{g}$ (Table 1). This value represents a remarkably high antioxidant activity when compared to several well-known antioxidant-rich foods, including elderberries $(10,655 \, \mu \mathrm{mol} \, \mathrm{TE} / 100 \, \mathrm{g})$, red apple $(7,781 \, \mu \mathrm{mol} \, \mathrm{TE} / 100 \, \mathrm{g})$, pomegranate $(5,923 \, \mu \mathrm{mol} \, \mathrm{TE} / 100 \, \mathrm{g})$, and plum $(5,700 \, \mu \mathrm{mol} \, \mathrm{TE} / 100 \, \mathrm{g})$ [16].
These findings highlight the significant potential of Collas-Co™ to combat oxidative stress through its high radical scavenging capacity. This exceptional antioxidant performance suggests its promising application in nutraceuticals and functional foods, where oxidative stress inhibition is essential for health benefits. Further studies exploring the specific bioactive compounds responsible for this activity could provide a deeper understanding of its mechanism and potential health applications.
# 3.2.2 DPPH Radical Scavenging Activity
The DPPH (2,2-diphenyl-1-picrylhydrazyl) assay is a commonly used method to evaluate antioxidant capacity [5]. It measures the ability of antioxidants to neutralize the stable DPPH radical, which has a deep violet color. When antioxidants donate electrons or hydrogen atoms, the DPPH radical is reduced to a pale-yellow or colorless compound, resulting in a measurable decrease in absorbance at $517~\mathrm{nm}$. This assay is valued for its simplicity, speed, and cost-effectiveness, making it widely used for screening antioxidant properties in natural products, foods, and supplements.
The DPPH radical scavenging activity of Gallic acid and Collas-Co $^{\mathrm{TM}}$ is presented in Fig. 1. Gallic acid exhibits moderate scavenging activity at lower concentrations, which increases steadily with higher concentrations. In contrast, Collas-Co™ shows relatively lower scavenging activity at lower concentrations but surpasses Gallic acid at higher concentrations. Notably, at the highest concentration (2.5 mg/mL), Collas-Co™ achieves a superior DPPH radical scavenging activity of 85.67%, outperforming Gallic acid, which achieves 70.66%, by approximately 15%.
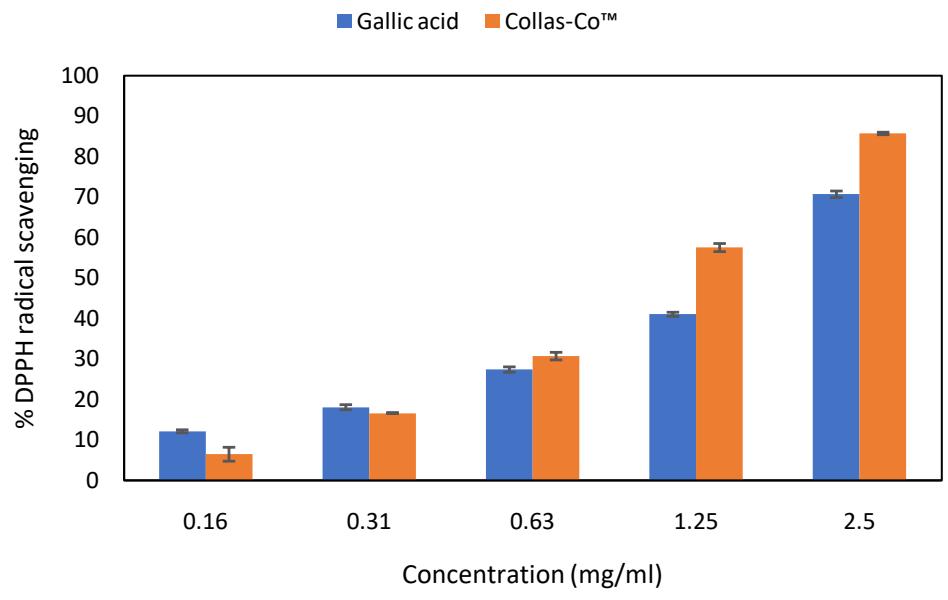
Fig. 1: DPPH radical scavenging activity of gallic acid and Collas-Co™
The IC50 value, a critical parameter for assessing the potency of a compound in inhibiting a specific biological or chemical process [17], is shown in Fig. 2 for the DPPH radical scavenging activity of Collas-Co™ and Gallic acid. The IC50 of Collas-Co™ is lower than that of Gallic acid, indicating that Collas-Co™ is approximately 21.8% more effective in achieving 50% inhibition of DPPH radical activity.
Although Gallic acid demonstrates higher radical scavenging activity at lower concentrations (as shown in Fig. 1), Collas-Co™ surpasses it in overall potency by requiring a lower concentration to reach the critical threshold of $50\%$ inhibition. This highlights Collas-Co™ as a more efficient scavenger at moderate to high concentrations. This result suggests that Collas-Co™ is better suited for applications requiring high efficacy at lower doses, making it ideal for products with concentration constraints. In contrast, Gallic acid remains a reliable and effective antioxidant option for formulations where higher concentrations are feasible and practical [18].
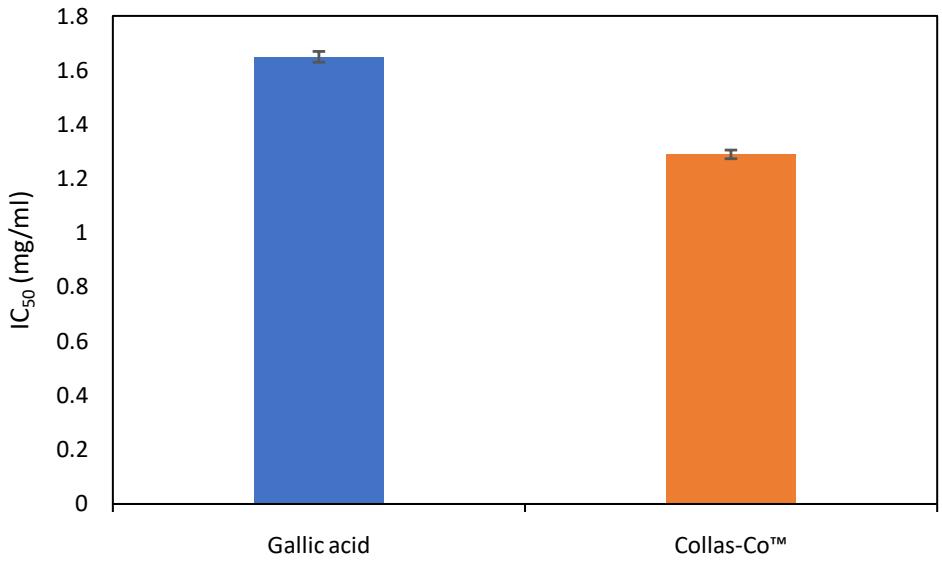
Fig. 2: Inhibition DPPH of gallic acid and Collas-Co™
# 3.3 Cytotoxicity Screening of Collas-Co™ on Primary Human Dermal Fibroblast
Fig. 3 presents an image of cultured human fibroblast cells observed under an inverted microscope. The inverted microscope offers a clear visualization of cell structures, allowing detailed assessment of cell health and morphology during experimental studies. Fibroblast cells exhibit a characteristic spindle shape with elongated, stretched-out bodies and occasional branching processes.
In the image, the cells grow in a monolayer formation, aligning parallel to one another, a hallmark of fibroblast behavior in vitro after 24 hours of culture. The parallel alignment and density of the cells suggest a confluent or semi-confluent layer, indicative of active growth and division under well-maintained culture conditions. The observed morphology and arrangement confirm that the fibroblast cells are healthy and thriving in optimal culture conditions, ensuring reliability for further experimental applications. This observation aligns with findings by D'Urso [6] in this study, which highlights the importance of mechanical and physical factors in maintaining fibroblast morphology and function under in vitro conditions.
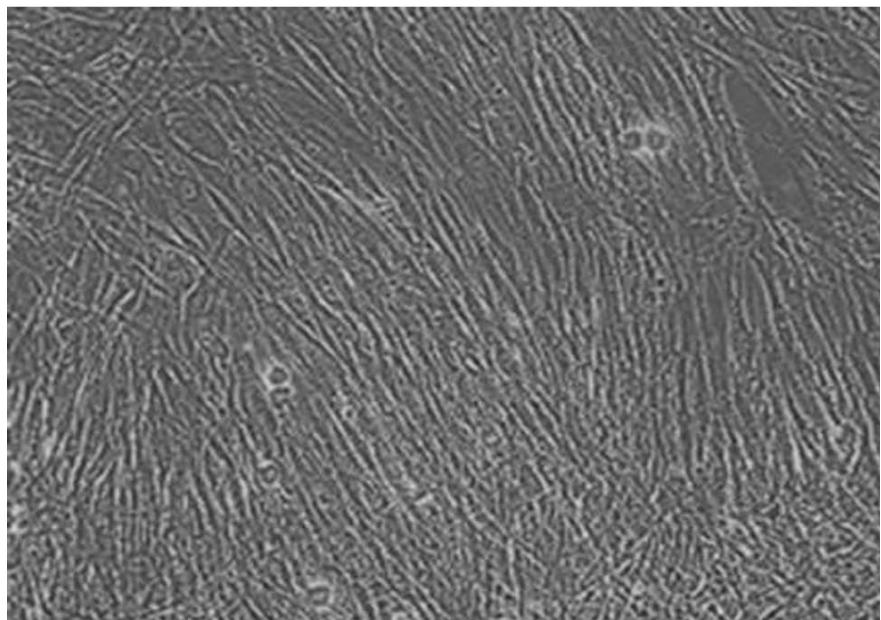
Fig. 3: Primary human Dermal Fibroblasts normal after aging for 24 hours
The cell viability of fibroblasts after 24 hours of exposure to various concentrations of Collas-Co™ and SDS is summarized in Table 2. Untreated fibroblasts in the control group maintained $100\%$ viability, providing a baseline for comparison. Exposure to $0.1\%$ Sodium Dodecyl Sulfate (SDS), a known cytotoxic agent, drastically reduced cell viability to $45.05 \pm 2.29\%$ , confirming its harmful effects. In contrast, Collas-Co™ showed a dose-dependent impact on fibroblast viability. At a low concentration of $0.39\mathrm{mg/mL}$ , cell viability increased to $112.94 \pm 2.20\%$ , indicating a protective or stimulatory effect. Moderate concentrations $(0.78 - 6.25\mathrm{mg/mL})$ led to a gradual decline in viability, although cells remained viable, suggesting tolerability within this range. At higher concentrations (12.50 - 50.00 mg/mL), cell viability significantly decreased, reaching $50.63 \pm 1.68\%$ at the highest concentration, highlighting dose-dependent cytotoxicity.
The $\mathrm{IC}_{50}$ value for Collas-Co™, calculated as 50.50 $\pm$ 2.07 mg/mL, indicates the concentration required to reduce cell viability by $50\%$ . This relatively high $\mathrm{IC}_{50}$ reflects moderate cytotoxicity, with stimulatory effects at lower concentrations and tolerability at moderate doses. The $\mathrm{IC}_{50}$ value, a critical parameter for assessing the potency of compounds, represents the concentration needed to inhibit $50\%$ of cell growth, with lower values indicating higher cytotoxicity. For comparison, a study revealed that the triple antibiotic paste (TAP)-cysteamine combination exhibited the lowest cytotoxicity (highest $\mathrm{IC}_{50}$ of $12.86\%$ ), indicating greater safety compared to other combinations such as chlorhexidine-cysteamine, which demonstrated the highest cytotoxicity with the lowest $\mathrm{IC}_{50}$ of $0.12\%$ [19].
Table 2: Cell viability of fibroblast after aging for 24 hours
<table><tr><td>Sample</td><td>Concentration</td><td>% Cell viability</td></tr><tr><td>Control</td><td>-</td><td>100.00 ± 0.00</td></tr><tr><td>SDS</td><td>0.1%</td><td>45.05 ± 2.29</td></tr><tr><td rowspan="8">Collas-Co™ (mg/ml)</td><td>0.39</td><td>112.94 ± 2.20</td></tr><tr><td>0.78</td><td>98.85 ± 2.62</td></tr><tr><td>1.56</td><td>93.31 ± 2.42</td></tr><tr><td>3.13</td><td>83.19 ± 2.70</td></tr><tr><td>6.25</td><td>75.56 ± 1.64</td></tr><tr><td>12.50</td><td>69.49 ± 2.77</td></tr><tr><td>25.00</td><td>61.38 ± 0.24</td></tr><tr><td>50.00</td><td>50.63 ± 1.68</td></tr><tr><td colspan="2">IC50(mg/ml)</td><td>50.50 ± 2.07</td></tr></table>
* SDS: Sodium Dodecyl Sulfate used for positive control
# IV. CONCLUSION
The study on Collas-Co™ reveals several significant findings regarding its bioactive properties and potential applications. First, it demonstrates a remarkably high total polyphenol content (TPC) and antioxidant activity, surpassing various foods renowned for their health benefits. This highlights its capability to combat oxidative stress effectively. Second, the DPPH assay confirms that Collas-Co™ has superior radical scavenging activity at higher concentrations compared to gallic acid, emphasizing its efficacy in antioxidant applications. Third, cytotoxicity testing on fibroblast cells shows that Collas-Co™ enhances cell viability at lower concentrations and exhibits tolerable effects at moderate doses, suggesting potential for safe use in nutraceuticals or cosmetic formulations. Additionally, its IC50 value indicates moderate cytotoxicity, with stimulatory effects at appropriate dosages. Overall, Collas-Co™ demonstrates significant promise as an ingredient in health-promoting products, warranting further exploration of its specific bioactive compounds and mechanisms.
# ACKNOWLEDGMENTS
The authors would like to express their sincere gratitude to the Central Laboratory (Thailand) Co., Ltd., for their invaluable support in providing advanced analytical services. Additionally, we extend our heartfelt thanks to the Expert Center of Innovative Herbal Products, Thailand Institute of Scientific and Technological Research (TISTR), for their expert assistance and state-of-the-art facilities that greatly contributed to the success of this research. Your contributions have been integral to achieving the objectives of this study.
Generating HTML Viewer...
− Conflict of Interest
The authors declare no conflict of interest.
− Ethical Approval
Not applicable
− Data Availability
The datasets used in this study are openly available at [repository link] and the source code is available on GitHub at [GitHub link].